

A title
Image Box text
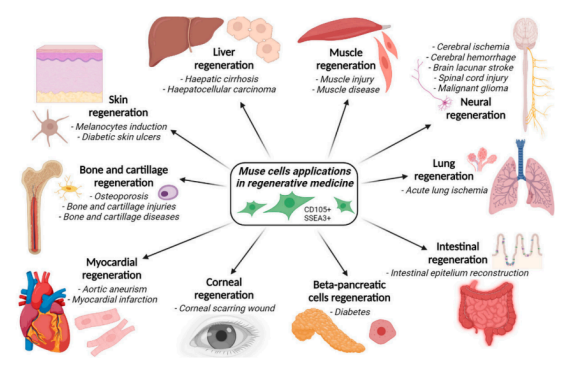
Muse Cells vs. MSCs: The Future of Regenerative Medicine
In the world of stem cell therapy, Mesenchymal Stem Cells (MSCs) have long dominated. They are sourced from bone marrow, fat, and umbilical cords. MSCs have been widely used in regenerative medicine. Though proven safe, they are helpful only for a limited number of health conditions, as they can only differentiate into specific cell types, such as bone, cartilage, and connective tissue cells.
So, science has been looking for something safe as MSCs, but much more potent. Science has long known that many such kinds of stem cells exist. And one such kind of much more potent cells are Muse cells — or Multilineage-differentiating Stress-Enduring cells.
Muse cells naturally occur in adult tissues and represent about 1–5% of MSCs. They’re small in number, but huge in potential.
Let’s explore why Muse cells are proving to be the elite choice in next-generation stem cell science.
What Makes Muse Cells Different?
Muse cells are pluripotent-like, meaning they can become nearly any cell type in the body. Unlike embryonic stem cells or iPSCs, they do not form tumors. They offer the regenerative power of embryonic cells, without the risk or controversy, and this safety have been proven in many clinical studies.
These cells also excel at:
Migrating to damaged tissues
Integrating into the target site

Replacing lost cells directly — not just supporting them

Avoiding immune rejection, thanks to HLA-G expression
These capabilities go far beyond what standard MSCs can achieve.
Side-by-Side Comparison
| Feature | Muse Cells | MSCs |
|---|---|---|
| Potency | Pluripotent-like | Multipotent |
| Tumor Risk | None | None |
| Homing to Injury | Guided by S1P signals | Often trapped in lungs |
| Immune Profile | HLA-G–mediated immune privilege | Moderate tolerance |
| Tissue Integration | High (via phagocytosis-driven repair) | Low (paracrine only) |
| Repair Duration | Long-term | Short-term support |
This is not to say that MSCs are far inferior, they have just fewer use case scenarios, they help in fewer health conditions. Muse cells have much broader impact.
How Muse Cells Work Smarter

1. They Go Where Healing Is Needed
Muse cells detect S1P, a distress signal released by damaged tissue. They migrate directly to the site of injury. Traditional MSCs? Often stuck in the lungs post-infusion.
In a heart attack model, Muse cells reached the heart and persisted for weeks, improving function.
2. They Become the Cells That Are Missing
Through a fascinating process called phagocytosis-triggered differentiation, Muse cells eat dying cells and read their signals. Then, they transform into the exact cell type needed to repair the damage.
In stroke models, Muse cells became neurons and restored motor function. MSCs only offered temporary support.

3. They’re Immune-Invisible
Muse cells express HLA-G, which protects them from immune attack. This means no HLA-matching or immunosuppressants are needed.
In clinical trials, patients received donor Muse cells without rejection. Cells survived and repaired tissues.
Real Results in Real Conditions
Heart Disease
In a 2022 study in animal models, Muse cells homed to infarcted heart tissue, became new heart muscle, and improved function — all without immune suppression.
In a 2020 human trial in Japan (CL2020), Muse cell-treated patients showed significant improvement in left ventricular ejection fraction within 3 months post-heart attack.
Stroke
In a 2023 randomized controlled trial, patients given Muse cells after stroke showed better recovery than placebo — with no adverse effects.

Spinal Cord Injury
In early trials, Muse cells helped some patients regain motor and sensory function. The cells were delivered IV — no surgery required.
ALS (Neurodegeneration)
In a 2023 study, ALS patients receiving monthly Muse infusions had a slower rate of decline than expected. The therapy was safe and well-tolerated.
Skin Repair (Wounds & Genetics)
In patients with epidermolysis bullosa, a rare genetic skin disorder, Muse cells helped close chronic wounds after just one IV dose.
In preclinical diabetic ulcer models, Muse cells promoted faster healing and formed real skin — not just scar tissue.
Above are just few examples, extensive studies are ongoing, these are just early days, and most studies already show safety and effectiveness of Muse cells therapy. Here it is also vital to note that many conditions in which muse cells are being tested are highly challenging to treat.
Muse Cell Exosomes: Healing Without the Cells?
Muse cells also release healing molecules, including:
Exosomes (tiny vesicles with RNA and proteins)

Secretome (a blend of cytokines and growth factors)
These may allow future cell-free therapies, using just the “messengers” Muse cells produce.
Studies show Muse secretome can reduce inflammation, prevent cell death, and promote tissue regrowth in heart, brain, and skin models.
Researchers are now exploring how to isolate and deliver Muse-derived exosomes in ways that could make regenerative medicine more accessible and less invasive.
Broad Applications. Singular Precision.
Muse cells are being tested across:
Cardiology (MI, heart failure)
Neurology (stroke, ALS, SCI)
Dermatology (wounds, genetic skin disorders)

Autoimmune diseases
Liver and kidney injury
In each field, Muse cells don’t just reduce symptoms. They aim to rebuild what’s broken.
Safety First
✅ No tumor formation observed in multiple long-term models
✅ Survive and function in the body for months
✅ No rejection even in unmatched donor recipients
✅ Extracted ethically from adult or umbilical tissues
While therapies are still investigational, the evidence is building. Multiple Phase 1/2 clinical trials are already completed or underway.
Why This Matters
For patients and physicians, Muse cells offer something traditional MSCs can’t:
Smarter navigation

Longer-term integration

Stronger functional recovery

Lower immune risk
The future of regenerative medicine isn’t just about stem cells. It’s about the right stem cells.
Muse cells are showing us what that future looks like — and it’s powerful.
Note:
Muse cell therapies are still investigational and not FDA-approved. Early clinical results are promising, but more large-scale trials are needed.
Stay informed. Stay curious. And watch this space.

Muse Cells in Regenerative Medicine:
What the Evidence Supports
A business-first review of clinical traction, risk profile, and commercialization relevance
(2019–2024)
I
Muse Cells in Regenerative Medicine: Recent Research
Muse cells (multilineage-differentiating stress-enduring cells) are described in the recent literature as a rare adult stem-cell subpopulation, identified around 2010, found across tissues such as bone marrow, blood, and skin, and typically estimated at roughly ~1% of the broader mesenchymal stem-cell pool. The central proposition is practical rather than theoretical: Muse cells aim to capture some “pluripotent-like” repair capacity while staying within an adult-cell safety profile, including an absence of tumor formation signals in early human datasets and limited immune friction in reported trials.

A title
Image Box text
1) Why Muse cells draw commercial interest
Pluripotent-like range, without the classic tumor narrative
Muse cells are reported to express pluripotency-related markers (SSEA-3, NANOG, OCT4, and others) and to differentiate into cell types spanning the three germ layers, while also showing biological constraints presented as reducing uncontrolled proliferation (low telomerase activity and high let-7 levels are cited in this context). From a product perspective, the logic is direct: if broad differentiation can be paired with a non-tumorigenic profile, the approach avoids one of the most common adoption barriers associated with embryonic-like platforms. Human
evidence here is still largely “no red flags observed so far,” rather than a definitive removal of long-term risk.
Off-the-shelf logic: immune tolerance as an operational advantage
The literature emphasizes an “immune-privileged” profile, often linked to HLA-G expression, and reports administration of HLA-mismatched donor Muse cells without immunosuppressants in the clinical programs described. If this continues to hold at scale, the implications are meaningful: standardized manufacturing, simpler
distribution, and a faster path to treatment in acute indications. The open question is how robust that tolerance remains across larger, more diverse populations and under repeated dosing.
2) Mechanistic positioning: coherent, not fully de-risked
Homing to injury via S1P gradients
A central mechanistic claim is that Muse cells respond to injury-associated sphingosine-1-phosphate (S1P) gradients, supporting migration from blood to injured organs more efficiently than conventional MSCs, which are commonly reported to be trapped in the lungs after IV infusion. This matters commercially because it strengthens the case for IV delivery as a scalable route and offers a clear differentiation story versus generic MSC therapies.
Phagocytosis-triggered “on-site” differentiation
Another distinctive mechanism described is phagocytosis-dependent differentiation: Muse cells engulf apoptotic debris at the damage site, and that process is reported to trigger differentiation aligned with the local cell types that were lost. This is a compelling biological narrative and fits the “smart repair” positioning, but the key uncertainty is how consistently and measurably this mechanism translates into durable functional integration in humans, beyond early observations.
Stress endurance and cellular resilience
Muse cells are also described as unusually stress-resistant, with robust DNA-repair and anti-apoptotic mechanisms. These claims support the idea that Muse cells could persist in hostile inflammatory environments, though the commercial relevance ultimately depends on whether this translates into reproducible clinical performance and predictable persistence profiles.

A title
Image Box text
A title
Image Box text
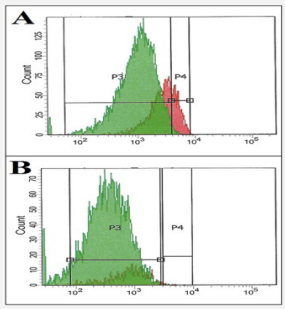
Muse cells generated insulin-producing cells more often than non-Muse cells (~10.1% vs ~3.9%). This difference is statistically significant (P<0.05), meaning it’s unlikely to be due to random variation in the experiment.
3) The current context of Muse cells
- Identity and biology. Muse cells are a small (<1 % of MSCs) subset of adult stromal cells identified around 2010 in bone marrow and connective tissues. They express pluripotency markers such as SSEA‑3, NANOG and OCT4 and can differentiate into derivatives of all three germ layers. Unlike embryonic or induced pluripotent stem cells, Muse cells have very low telomerase activity and intrinsic tumor-suppressor mechanisms (e.g. high let‑7 microRNA expression), along with robust DNA‑repair pathways. These properties are associated with stress tolerance and absence of teratoma formation, making Muse cells attractive for regenerative medicine.
- Migration to injury. Muse cells express receptors for sphingosine‑1‑phosphate (S1P); they mobilize from the bone marrow into peripheral blood and home to damaged tissues that release S1P as a distress signal. Clinical observations have shown that patients with acute myocardial infarction or stroke exhibit increased circulating Muse cells in the acute phase, correlating with subsequent recovery. This suggests endogenous Muse cells are recruited as an innate repair mechanism following tissue injury
- Immune privilege. Muse cells display low immunogenicity and immune‑privileged characteristics. Notably, they express HLA‑G (a non-classical HLA associated with fetomaternal tolerance) and produce immunosuppressive factors like IDO, which likely enable allogeneic Muse therapy without systemic immunosuppression. Donor Muse cells have been shown to survive and differentiate in host tissues for months without rejection. They also secrete anti‑inflammatory, anti‑apoptotic and tissue‑protective factors that modulate the local microenvironment.
Key research findings (2019–2024)
Cardiovascular indications
Ovine model of acute myocardial infarction (AMI)
- Study design: Adipose‑derived Muse cells (AT‑Muse) were isolated and administered intramyocardially to sheep with induced AMI. A 2023 PLOS One study reported that AT‑Muse cells expressed higher angiogenic factors (VEGF, PGF, ANG) and formed more capillary-like tubes in vitro than conventional adipose-derived MSCs.
- Outcomes: In treated sheep, histology at one week post-infarct showed significantly more arterioles and capillaries in the infarct border zone compared to vehicle controls. Injected Muse cells (tracked via PKH26 labeling) were found co-localized with cardiac markers (desmin, sarcomeric actin, troponin T) and an endothelial lectin, indicating differentiation into cardiomyocyte and vascular lineages. The authors concluded that intramyocardial Muse cell administration significantly promoted neovascularization and cell survival in the ovine AMI model.
- Implications: These pre‑clinical findings support the pro‑angiogenic, tissue-integrating behavior of Muse cells in cardiac repair. However, the study had a small sample size, and translation to humans requires careful dose-scaling and safety assessment.
First-in-human trial in AMI
- Study design: A first-in-human pilot trial in Japan (2020) administered an allogeneic Muse cell product (CL2020, 1.5×10^7 cells IV) to 3 patients with acute ST-elevation MI and left ventricular ejection fraction (LVEF) ≤45% after PCI. Cells were given intravenously (leveraging their homing ability) within days post-infarction, and patients were followed for 12 weeks.
- Outcomes: No adverse drug reactions or safety issues were observed following Muse cell infusion. Treated patients showed marked functional improvements: LVEF and wall-motion scores improved over the 3-month follow-up period. This contrasts with the minimal spontaneous recovery typically seen after MI in that timeframe.
- Implications: Although uncontrolled and very small (n=3), this trial provided initial human evidence that systemically delivered Muse cells can safely enhance cardiac functional recovery after MI. It demonstrated the feasibility of IV administration (a less invasive route than intracoronary injection) and suggested potential efficacy. A larger, controlled trial will be needed to confirm these benefits and determine the degree of improvement attributable to Muse cells
Neurological indications
Stroke (review and pre‑clinical evidence)
- Limitations of existing therapies: A 2022 review highlighted that the only FDA‑approved therapy for acute ischemic stroke (tissue plasminogen activator, tPA) is administered to <5 % of patients due to a narrow time window, and many neuroprotective agents have failed clinically. This underscores a large unmet need for therapies that can aid recovery in the subacute phase.
- Advantages of Muse cells: Muse cells are naturally pluripotent, home to damaged brain tissue, survive harsh ischemic conditions and spontaneously differentiate into neurons and glia. The review summarized that in SCID mouse stroke models, human Muse cells injected intravenously migrated to peri‑infarct regions, differentiated into neural cells (Tuj‑1^+ neurons, NeuN^+ mature neurons), survived longer than non‑Muse MSCs, and improved functional recovery. In a lacunar stroke model, Muse cells promoted greater behavioral recovery than unsorted MSCs and did not form tumors.
- Logistics and cost: The review noted that cryopreservation and consistent culture conditions are needed for clinical use; however, manufacturing Muse cells at scale poses challenges. Yields are lower and culture costs higher than for bulk MSCs, raising CMC (chemistry, manufacturing, control) hurdles for widespread use.
- First clinical trial evidence: In late 2023, a randomized double-blind trial in Japan evaluated intravenous allogenic Muse cells for subacute stroke. Thirty-five ischemic stroke patients 2–4 weeks post-onset were randomized to a single infusion of Muse cells (n=25) or placebo (n=10) without immunosuppressants. At 12 weeks, 40% of Muse-treated patients achieved significant functional recovery (mRS ≤2) versus 10% in the placebo group. This met the pre-specified efficacy threshold and, despite the small sample, suggests a meaningful improvement in disability outcomes. Adverse events were common in both groups (related to underlying stroke), and one serious event (a transient seizure) occurred in the Muse arm. Overall, the trial demonstrated that Muse cells are a possibly effective and tolerable treatment for subacute stroke, warranting larger trials for confirmation.
Amyotrophic lateral sclerosis (ALS)
- Preclinical rationale: ALS is a neurodegenerative disease with no cure, and prior mouse studies indicated Muse cells might protect motor neurons. In SOD1-transgenic ALS mice, intravenously delivered human Muse cells homed to the spinal cord and significantly improved hindlimb strength in repeated-dose experiments. These cells also modulate inflammation (e.g., reducing microglial activation), providing a scientific basis to test Muse therapy in ALS.
- Phase 2 trial: A 2023 single-center trial in Japan explored the safety and feasibility of repeated Muse cell
infusions in ALS patients. Five patients received six monthly doses of CL2020 (each 1.5×10^7 cells IV). The primary
endpoints were safety and tolerability. Safety: The treatment was well tolerated; no infusion-related serious
adverse events occurred over 12 months. Efficacy signals: Although the trial was open-label and not powered for
efficacy, 3 of 5 patients showed a slower decline in ALSFRS-R functional score after Muse therapy, compared to
their pre-treatment trajectory. The overall ALSFRS-R slope post-treatment trended favorably (slower disease
progression) relative to the pre-treatment period, but due to the small sample it did not reach statistical
significance. Inflammatory biomarkers (IL-6, TNF-α) rose transiently post-infusion, and neurodegeneration markers
(NfL, CHIT1) also increased, which may reflect disease activity or immune engagement. Implications: This early trial
suggests repeated dosing of Muse cells in ALS is feasible and safe. There were indications of a potential slowing of
disease progression in some patients, but given the uncontrolled design and variable responses, efficacy remains
unproven. A larger placebo-controlled trial is planned to determine if Muse cells can meaningfully alter ALS
outcomes.
Gastrointestinal syndrome
Radioprotection and intestinal regeneration
- Fetal Muse-based therapy: A 2023 study (Stem Cell Research & Therapy) evaluated a single intravenous injection of umbilical cord–derived Muse cells (WJ‑Muse) after lethal abdominal irradiation in mice. Key outcomes: the treatment significantly improved survival by regenerating intestinal epithelium and restoring barrier function. Early increases in IL‑6 and MCP‑1 promoted recruitment of monocytes/M2 macrophages, and stimulated Paneth cell proliferation via IL‑6/Stat3 signaling. The authors concluded that a single WJ‑Muse injection could provide a simple therapeutic strategy for lethal gastrointestinal syndrome (GIS).
- Context: This study emphasized that Muse cells migrate to injured tissues, exert anti‑inflammatory, anti‑apoptotic and vascular protective actions, and evade host immune rejection. It also noted ongoing clinical trials of Muse therapy for cardiac, neurological, spinal cord injury and ALS indications, indicating broad translational potential.
- Limitations: The GIS model used immunodeficient mice; translational relevance to humans will require safety evaluation and dosing optimization in large-animal models or clinical trials. No human data in acute radiation syndrome are yet available.
Skin and cluster biology
Muse cell cluster properties
A 2023 study examined how sorted Muse cells form clusters (“M-clusters”) in suspension culture. Key findings: magnetically sorted SSEA3^+CD105^+ cells formed sphere-like clusters with ~87% purity; clusters stained positively for alkaline phosphatase, SSEA‑3 and OCT4, and had outer membranes composed of amorphous material. The authors concluded that MACS‑sorted Muse cells can generate multi-cell clusters containing cells of varying stemness wrapped in membrane-like structures. This provides insights into Muse cell culture and enrichment methods, which could be relevant for scaling up manufacturing.
Dystrophic epidermolysis bullosa (EB) trial
- Pilot study: Muse cells have been tested in a severe genetic skin disease, recessive dystrophic EB, characterized by non-healing wounds. A phase 1/2 open-label study in 2021 infused a single dose of CL2020 Muse cells (1.5×10^7 IV) into 5 adult EB patients with chronic refractory skin ulcers. Patients were observed for 52 weeks post-infusion.
- Outcomes: All patients tolerated the infusion without serious toxicity; only mild, self-limited events were reported (e.g. one Grade 3 abdominal pain, transient fever, mild infections). By 4 weeks post-treatment, 2 of 5 patients achieved >50% wound area reduction, and the mean ulcer size across all patients had decreased by ~46%, a statistically significant improvement from baseline. Some wounds showed rapid epithelialization (one patient’s ulcers healed fully at 4 weeks). Patients also reported reduced pain at the ulcer sites in the early weeks. However, the benefit appeared temporary – by 3 months, most ulcers had returned to roughly their original size as skin fragility persisted. No increase in collagen VII or new anchoring fibrils was detected on biopsy, indicating that the fundamental genetic defect was not corrected.
- Implications: This first-in-human application in a dermatological condition showed that Muse cells can be delivered safely and may induce transient wound healing even in a challenging genetic disorder. The short-lived response suggests that repeated dosing or higher cell numbers might be needed for sustained healing in EB. Nonetheless, the trial provided proof-of-concept that Muse cells can engraft in injured skin and promote tissue repair without immunosuppressive therapy (all patients received unmatched donor cells and none experienced rejection). Further studies in chronic wound conditions could explore optimized dosing schedules to maintain the regenerative effect.
Translational considerations
- Efficacy signals: Across diverse pre‑clinical models, Muse cells have shown the ability to promote tissue regeneration: improving neovascularization in an infarcted heart, enhancing functional recovery in stroke models, rescuing GI mucosal integrity after radiation, and accelerating wound healing in skin ulcers. These effects are achieved through a combination of differentiation into host tissue cells and robust paracrine/immunomodulatory actions that create a pro-regenerative milieu.
- Safety profile: Muse cells have not produced teratomas or triggered immune rejection in animal studies. Notably, recent human trials likewise reported no tumorigenic events and did not require HLA matching or immunosuppression for allogeneic Muse cell administration. This immune-privileged behavior, attributed partly to HLA-G expression, differentiates Muse cells from embryonic stem cells and iPSCs, which carry tumor risk and immunogenicity. The absence of graft-versus-host issues in multiple Muse trials (stroke, ALS, EB, MI) is a key advantage for clinical deployment.
- Manufacturing risks: Muse cells constitute only ~1 % of an MSC population; isolating and expanding them to therapeutic doses requires specialized methods (e.g. cell sorting, cluster culture). Yields are limited, and culture conditions must preserve their unique properties. As a result, production costs are higher than for bulk MSCs, and batch-to-batch variability in quality could limit scalability. Ensuring consistent SSEA-3^+ cell recovery and viability at scale remains a challenge for commercialization.
- Regulatory landscape: Muse cell therapy is still in early translational stages. Without large-scale, randomized clinical trials demonstrating consistent benefit and safety, regulatory approval will be challenging. For example, the encouraging stroke trial result will need confirmation in a Phase 3 study before any approval. The time-sensitive nature of conditions like stroke also implies that an off-the-shelf, cryopreserved Muse cell product is necessary for real-world use. Developers must also navigate intellectual property (the Muse cell concept is patented) and ensure freedom-to-operate, as well as meet stringent CMC and quality control requirements
II
Clinical Trials of Muse Cells (Multilineage-Differentiating Stress-Enduring Cells)
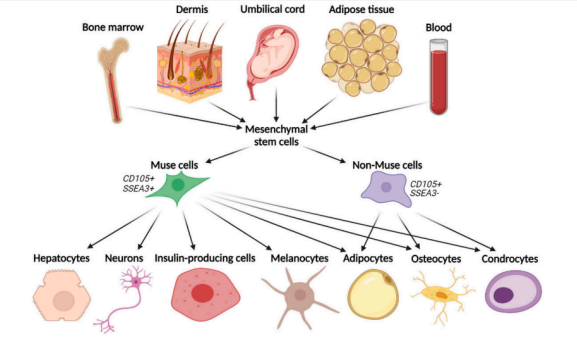
Graphical depiction of tissue origin and differentiation capacity of Multilineage–differentiating stress–enduring cells (Muse cells).
Cardiovascular: Acute Myocardial Infarction
First-in-human pilot (Japan, 2018; published 2020)
This initial human program targeted acute STEMI patients with reduced LVEF after PCI. Three patients (LVEF ≤45%) received a single intravenous dose of 1.5×10^7 allogeneic Muse cells (CL2020), with follow-up over 12 weeks. The reported signal was straightforward: no infusion-related safety issues were observed, and LVEF improved materially versus baseline, with changes described as exceeding 10 percentage points on average, alongside improved wall motion scores. The framing in the source material emphasizes that such a jump in LVEF is considered large in the context of heart failure therapies, and that arrhythmias or other major safety complications were not seen during follow-up. The practical conclusion from this early work is limited but clear: feasibility was demonstrated, acute tolerability looked acceptable, and the direction of functional change supported continued development.
Observational support: endogenous Muse mobilization after MI
In parallel, observational data supports the biological premise that Muse cells participate in injury response. An observational study in 79 AMI patients reported that circulating SSEA-3+ Muse cell counts rose and peaked around 24 hours post-infarct, in parallel with elevated S1P levels described as a chemotactic stress signal. A larger “surge” of Muse cells in the acute phase was associated with better left ventricular function and less remodeling at six months. The business-relevant point is not that this proves efficacy of infusion, but that it offers a coherent story for mechanism plausibility and patient stratification logic, while also giving a rationale for why timing (early window, stress-signal gradient) could matter operationally in future trial designs.
Neurology: Ischemic Stroke (Subacute Phase)
Phase 2 randomized controlled trial (Japan, 2018–2023)
The stroke dataset is the most decision-useful in this set because it includes a placebo-controlled structure. The trial (JapicCTI-184103) tested intravenous CL2020 in subacute ischemic stroke patients treated 2–4 weeks after stroke onset. Thirty-five patients with moderate-to-severe deficits were randomized to a single IV infusion of Muse cells (n=25) or placebo (n=10), without immunosuppressants. Safety was handled as the primary endpoint at 12 weeks, and the sources describe no safety “show-stoppers.” Adverse events were common in both groups, which is not surprising in a stroke rehabilitation population; the key detail is that serious events judged related to the product appear limited (a seizure is singled out as related in the text), and there is no claim of excess severe adverse event burden versus placebo.
Efficacy signals were framed using functional independence thresholds. At 12 weeks, 40% of treated patients reached mRS ≤2 versus 10% in placebo. At 52 weeks, response rates were described as 68.2% versus 37.5%, with a further note that mRS 1 (near-complete functional recovery) was achieved in about one-third of treated patients, while none of the placebo group reached mRS 1 within a year in the cited summary. Taken at face value, this is a clinically meaningful direction of effect, with durability suggested by the one-year follow-up, but the usual constraints remain: sample size is modest, allocation is uneven, and replication at scale becomes the next gating step. Still, within this trial set, stroke reads as the closest thing to a proof-of-concept that goes beyond safety into a controlled efficacy signal.
Neonatology: Hypoxic-Ischemic Encephalopathy
Phase 1 open-label dose-escalation (Japan, 2019–2021)
The HIE program (“SHIELD”) was an investigator-initiated open-label safety study in newborns already receiving therapeutic hypothermia. Nine neonates with moderate-to-severe HIE were enrolled using a 3+3 style dose-escalation. Each infant received a single IV infusion of CL2020 between 5 and 14 days of age, at either 1.5×10^6 cells (n=3) or 1.5×10^7 cells (n=6).
The reported safety picture is clean in the context of a fragile population: no infusion reactions or clinically significant physiologic instability during/after infusion, with only a transient mild elevation in one liver enzyme in one infant described as possibly related and self-resolving. All nine infants survived, and no serious adverse events were attributed to the cells.
Early outcome framing is cautious because the study is uncontrolled, but the text reports that by 18 months, 67% of treated infants had normal neurodevelopmental scores across domains on the referenced scale. The value of this dataset is primarily that it removes some of the “unknown unknowns” around acute tolerability in neonates and supports moving to a controlled design, while also showing explicit linkage to the patent ecosystem through author disclosures and product sourcing.
Neurotrauma: Cervical Spinal Cord Injury
Phase 1 open-label multicenter (Japan, ~2020–2022)
This program enrolled ten patients with cervical SCI (levels C4–C7, incomplete paralysis grades B1/B2) in a prospective single-arm study (JRCT1080224764). Each received a one-time IV infusion of 15×10^6 CL2020 Muse cells, following regulatory review.
Safety is again the lead claim: no serious safety issues emerged, and severe adverse events reported during follow-up were not deemed related to therapy. The sources emphasize tolerability without immune complications and no requirement for immunosuppression.
Because the design is uncontrolled, efficacy statements need to be treated as signals rather than conclusions; still, the investigators reported improvements in motor scores, activities of daily living, and quality-of-life indices compared with baseline. The business takeaway is that SCI sits in the “encouraging feasibility + signal, but not yet decision-grade efficacy” bucket, and the sponsor alignment is consistent with a patent-holder-driven pipeline strategy.
Neurodegeneration: Amyotrophic Lateral Sclerosis
Phase 2a open-label repeated dosing (Japan, 2021–2023)
ALS is notable here because it uses a repeated-dosing regimen, which matters operationally for manufacturing, logistics, and long-term safety. The trial (jRCT2063200047) enrolled five patients and administered six monthly IV doses of allogeneic Muse cells (CL2020), one infusion every four weeks for six months
Safety is described as favorable, with no serious adverse effects reported, and the repeated infusions did not trigger acute reactions or long-term safety issues in the small sample. Clinical outcomes were tracked using ALSFRS-R trends, and the text describes a slowing of decline in most patients versus the pre-treatment run-in period, with variability across individuals and no statistical significance due to sample size. Biomarker commentary is included in the source material, describing transient changes in inflammatory markers and a steady decline in serum S1P over 12 months, positioned as potentially relevant to homing biology, while remaining interpretive.
The practical conclusion stays conservative: this is a safety and feasibility readout with a suggestive disease-modifying signal that is not strong enough to price as efficacy without a controlled follow-on trial. The explicit note that inventors on the study hold patents in ALS indications reinforces the “patent-linked trials” criterion guiding the selection here.
Dermatology: Epidermolysis Bullosa
Phase 1/2 open-label (Japan, 2018–2021)
The EB dataset involves five adult RDEB patients with chronic refractory ulcers receiving a single IV infusion of 1.5×10^7 CL2020. Safety at 12 weeks was the primary endpoint; adverse events were largely mild and transient in the summary, with one Grade 2 lacrimal duct stricture noted and managed without major complications, and only mild transient paresthesia described as possibly related.
Efficacy signals in an uncontrolled setting were framed around wound area reduction, with two of five patients achieving >50% reduction in at least one target ulcer by four weeks, and an overall decrease in tracked ulcer area across 52 weeks. Patient-reported pain reduction and improvements in inflammatory markers were also mentioned. From a commercialization viewpoint, EB is attractive because endpoints can be visually measurable and clinically meaningful even in small cohorts, but the current evidence remains early-phase and single-arm, so the next step would be a design that better separates treatment effect from wound variability and supportive care effects.
Pulmonary: ARDS (COVID-19 era)
Exploratory program (Japan, 2020)
A Muse cell trial was initiated for COVID-related ARDS (jRCT2043210005), sponsored by Life Science Institute. The stated intent was to assess safety in severe inflammatory lung injury and explore whether immunomodulatory and reparative properties could translate into clinical benefit. As described, detailed results were not fully published as of the stated timeframe, and the available commentary is closer to early reporting than a formal dataset, with anecdotal indications and emphasis on absence of serious safety issues in a small experiment. The correct posture here is to treat ARDS as a pipeline extension attempt with limited disclosed outcomes to date, rather than a program with mature clinical evidence.
Cross-trial readouts that matter in practice
1) Safety and immune handling are the consistent theme
Across these indications, the sources repeatedly emphasize tolerability, lack of major infusion reactions, and the operational feature that dosing was generally done without HLA matching or immunosuppression, positioned as linked to immune-evasive characteristics such as HLA-G expression. Even allowing for early-stage bias and small samples, the consistency across organs and patient groups supports the view that the core development strategy is built around an “off-the-shelf allogeneic” narrative with manageable short-term safety in the cohorts tested so far.
2) The strongest efficacy-grade signal sits in stroke, mainly because of the control arm
When the evidence is filtered for designs that can plausibly separate signal from noise, the stroke randomized trial stands out. The AMI pilot, HIE, SCI, ALS, EB, and ARDS programs are informative, but the absence of control arms (or very small sample sizes) makes the magnitude and durability of benefit difficult to price confidently. Stroke provides a cleaner comparison and a longer follow-up readout, which is why it tends to carry more weight in an investment-style review, even though replication remains necessary.
3) Dosing strategy is still not settled across indications
Most programs used a single infusion at roughly similar dose levels (often 1.5×10^7 cells), while ALS tested monthly repeat dosing and remained small. That divergence matters because a therapy that requires repeated dosing moves the economics and operational footprint materially, especially if manufacturing throughput becomes a limiting factor. In the current dataset, repeat dosing looks feasible from a safety standpoint in a very small sample, but it does not yet answer durability questions at scale.
4) Patent linkage is visible and not accidental
The selection criterion was alignment with the patent-holding ecosystem, and the text supports that linkage. Many trials are led or funded by the same groups tied to the Muse cell IP, including Life Science Institute sponsorship and author patent disclosures in several studies. In practice, this matters because it suggests the clinical pathway is being used to operationalize patent claims into specific indications, and that the “trials that matter” are not generic academic curiosity; they are part of a controlled development program around a proprietary product.
5) The obvious gaps are scale, replication, and publication completeness
The overall pattern is familiar for early regenerative medicine programs: small cohorts, single-arm designs in several indications, and a few controlled datasets that show directionally positive results. The next value inflection in this story will come from larger, controlled trials that can confirm effect sizes, define responder profiles, and standardize endpoints across centers, while also clarifying how outcomes behave outside Japan-based systems and investigator networks. The ARDS example also highlights that some initiatives exist but remain thinly published, which adds uncertainty until full datasets are disclosed.
III
Conclusions about the future outlook
Strength of evidence
The past five years have yielded promising evidence that Muse cells can home to injury, integrate into tissues and aid functional recovery. Importantly, initial human clinical data have emerged: a small controlled trial in stroke showed improved outcomes over placebo, and pilot studies in MI, ALS, and EB demonstrated safety and hints of efficacy. However, most data still derive from small animal models or uncontrolled human series. Only a single randomized trial to date (with 25 treated patients) has tested efficacy, and overall patient numbers remain very limited. Thus, while the therapeutic concept appears plausible and offers advantages over general MSC therapy, clinical efficacy and scalability remain unproven.
Remaining gaps and risks
- Clinical translation: Robust, adequately powered human trials are needed to confirm benefits in MI, stroke and other indications. The existing stroke RCT result is encouraging but needs replication in larger Phase 3 trials. Long‑term follow-up is necessary to assess the durability of Muse cell engraftment and any late-onset adverse events (e.g., ectopic tissue formation or autoimmune reactions) that might not appear in short studies.
- Manufacturing: Standardizing isolation and expansion protocols will be critical for reproducibility. Variability in cell yield or potency could undermine trial results. Developing a centralized manufacturing process (e.g., a bioreactor or automated sorting system for Muse cells) may be required to generate consistent product for multicenter trials. Enhancing the proportion of Muse cells or efficiently producing them from allogeneic donors (perhaps by leveraging the cluster culture method) is an active area of research.
- Cost of goods: The complexity and low throughput of Muse cell production raise the cost of goods significantly above that of conventional MSC therapies. Unless Muse cells demonstrate markedly superior efficacy, these higher costs may limit commercial viability or relegate Muse therapies to niche, high-need indications. Economic considerations will factor into development decisions, especially if repeat dosing is required (as suggested in ALS and possibly EB).
- Regulatory acceptance: Regulators will require rigorous evidence of safety, consistency, and clinical benefit. The unique aspects of Muse cells (e.g., spontaneous differentiation in vivo, heterogenous clusters) will need clear characterization in an IND/BLA filing. Any proprietary claims around Muse cells could face patent challenges; companies pursuing Muse-based products must ensure freedom-to-operate or obtain licensing (e.g., from the Japanese groups that pioneered the platform). Overall, without clear superiority in outcomes, convincing regulators and payers of Muse cells’ value proposition could be difficult in a crowded regenerative medicine field.
Summary
Significant progress has occurred since Muse cells entered the scientific discussion around 2010. The core scientific positioning remains consistent across the literature: Muse cells express pluripotency-associated markers such as SSEA-3, Oct 3/4, NANOG, Sox2, and Par-4, but generally at lower levels than embryonic stem (ES) or induced pluripotent stem (iPS) cells. The practical implication is that Muse cells sit in a middle zone, with broad differentiation potential paired with a safety profile that is presented as closer to adult-cell platforms.
Mechanistically, Muse cells are reported to differentiate into all three germ layers in vitro and in vivo without teratoma formation, and to maintain a stable, normal karyotype in culture (normal chromosome number and integrity). The literature also frames naïve Muse-AT cells as quiescent, which is used to explain their resilience during transplantation and their ability to survive in hostile injury environments. Taken together, this is the reason Muse cells continue to attract attention: broad repair capacity, combined with claims of lower tumor risk and manageable immune friction.
The efficacy story is still anchored primarily in preclinical evidence, but it is not trivial. Across animal disease models, Muse cells are reported to home to sites of injury, integrate into damaged tissue, replenish cell populations, and contribute to functional recovery in multiple organs, including skin, muscle, liver, kidney, and neural tissues. That breadth is both an opportunity and a risk: it supports a platform narrative, but it also raises the bar for proof in humans, where biology tends to be less forgiving and endpoints are harder.
On the evidence-quality side, the recent PMC-based search captured prominent open-access studies up to 2024, but it should be treated as directional, not complete. Relevant work may sit outside open-access channels, including late 2024 to 2025 publications, conference abstracts, proprietary datasets, or industry reporting. For due diligence purposes, the next layer of work should widen coverage through larger indexing platforms (e.g., Scopus, Web of Science), and should systematically track registered clinical trials and any disclosed readouts. This is the most direct way to measure whether the field is compounding or stalling.
From a business perspective, the main bottlenecks are predictable: manufacturing yield, scalability, consistency across batches, and reliable delivery to the right tissue at the right dose. The research direction most likely to unlock value is not only “more indications,” but better control of the product: improving Muse cell yield, refining expansion protocols, and strengthening delivery methods (including bioengineering approaches that improve targeting or persistence). These are the types of technical advances that can turn an interesting biology story into a more durable clinical and commercial asset.
Overall, Muse cells still look like a platform in early formation, not a finished product. The upside case rests on a combination that is rare in regenerative medicine: a credible safety narrative, a differentiated mechanism of action (homing, integration, and repair), and the potential for an off-the-shelf treatment model. The cautious view is that the evidence base, while encouraging, remains uneven across indications and still needs larger, controlled human datasets to define real effect sizes. The most sensible stance is selective optimism: Muse cells are worth watching closely, with attention focused on concrete inflection points, including Phase 2/3 readouts, manufacturing breakthroughs, and consistency of outcomes as trials expand beyond small cohorts and single-center settings.
References
pubmed.ncbi.nlm.nih.gov (biomedical abstracts database)
startstemcells.com (secondary science summary site)
researchgate.net (researcher shared publications)
academic.oup.com (OUP journals platform)
rpip.tohoku.ac.jp (Tohoku IP office pages)
patentimages.storage.googleapis.com (patent PDF image host)
nature.com (scientific journal publisher)
frontiersin.org (open-access journals publisher)
medcraveonline.com (open-access journal site)
pmc.ncbi.nlm.nih.gov (full-text biomedical archive)
jidonline.org (Journal of Investigative Dermatology)
iris.univr.it/retrieve/e527bbc8-5480-491d-8778-11c6e3722a97/biomedicines-11-01587-v2-1.pdf (Academic journal)
sciencedirect.com/science/article/pii/S1687157X18300842 (Academic journal)
