
A title
Image Box text
Disclaimer
Mesenchymal stem cell (MSC) therapy is an evolving field of regenerative medicine. While numerous studies demonstrate encouraging biological effects, not all MSC applications for chronic diseases are yet fully approved by the U.S. Food and Drug Administration (FDA). Current use should be considered as supportive, not replacing, standard medical care. Patients should discuss any stem cell–based treatment with qualified healthcare professionals and continue all prescribed therapies.
Of course, this does not mean that MSC does not work. After all, it is not some unproven herbal remedy or some mysterious treatment. Nonetheless, it is vital to understand that it has still not been fully recognized, thus it is not included in many guidelines. It is still being tested in many health conditions with some excellent early results.
With that said, also remember that stem cell therapy offers great hope! Medications can help manage chronic diseases, while stem cell therapy offers hope of a cure. It may completely reverse certain health issues so that people can get rid of chronic diseases. Thus, for example, stem cell therapy may enable the pancreas to start producing insulin again in those living with type 1 diabetes. So, here we look at what to expect from the therapies we offer.
Stem Cell Therapy and Its Emerging Role in Chronic Disease Management
Chronic diseases such as diabetes, cardiovascular disease, arthritis, autoimmune disorders, and metabolic syndromes share common features: persistent inflammation, progressive tissue damage, impaired repair mechanisms, and declining function.
MSCs are under investigation because they offer immunomodulatory, anti-inflammatory, and regenerative potential, which may help shift the body’s repair balance. For instance, a recent review, published in a highly reputed journal (Nature), states that MSCs are “a highly promising strategy in regenerative medicine … across a broad spectrum of human diseases”.
Below, we break down major disease categories, summarize clinical evidence (with hyperlinks), note caveats, and highlight what the current research suggests.
1. Diabetes & Metabolic Disorders
Medications can manage diabetes, but none can cure or reverse the condition. Therefore, regenerative medicine offers great hope. At present, it can help partially reverse certain degenerative changes caused by diabetes, thus helping reduce dependence on medications.
Also, keep in mind that when living with diabetes, stem cell therapy can also help prevent and manage certain diabetes related complications, or at least, delay their development significantly.
In both type 1 and type 2 diabetes mellitus (T1DM, T2DM), the key challenges include β-cell loss (T1), insulin resistance (T2), microvascular damage, persistent inflammation, and complications such as foot ulcers.
What the evidence shows:
A systematic meta-analysis: “Efficacy of mesenchymal stem cell therapy on glucose levels in type 2 diabetes” saw significantly reduced HbA1c after MSC therapy.
A recent human review: “Human mesenchymal stem/stromal cell-based therapy in diabetes mellitus” summarizes outcomes in T1 & T2. This review states that “…MSC can ameliorate peripheral insulin resistance, halt beta-cell destruction, preserve residual beta-cell mass, promote beta-cell regeneration and insulin production, support islet grafts, and correct lipid metabolism.”
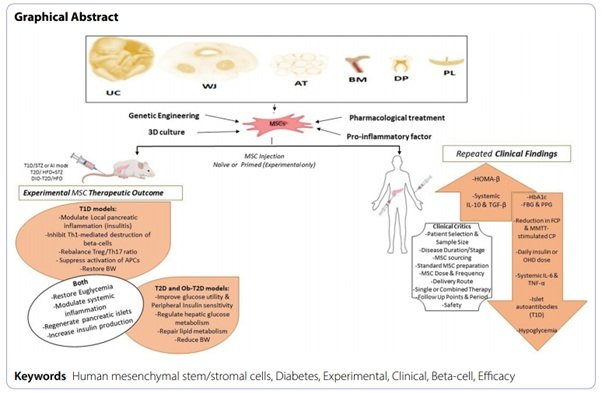
Figure 1 Stem cell therapy/MSC-therapy in diabetes (CC-BY image. Image source: Zeinhom, A., Fadallah, S.A. & Mahmoud, M. Human mesenchymal stem/stromal cell based-therapy in diabetes mellitus: experimental and clinical perspectives. Stem Cell Res Ther 15, 384 (2024). https://doi.org/10.1186/s13287-024-03974-z)
Clinical implication: MSC therapy may reduce insulin-dose requirements, improve glycemic control, and assist complication healing (e.g., foot ulcers) — but does not replace insulin or oral therapies.
So, if you are living with diabetes and experiencing diabetes related complications, MSC therapy might be an option.
2. Cardiovascular & Peripheral Vascular Disease
Cardiovascular conditions are the number one cause of mortality globally. These diseases occur due to multiple reasons, like certain dietary choices, smoking, alcohol intake, stress, genetics, and much more. Stem cell therapy or MSC-therapy cannot fully cure. Moreover, by the time these conditions are diagnosed, much damage has already occurred. Nonetheless, MSC-therapy, when used as a part of a treatment complex, can benefit significantly in some instances.
Ischemic injury, poor perfusion, fibrosis, and loss of functional tissue underlie many cardiovascular disorders and peripheral vascular disease. MSCs offer angiogenic, anti-fibrotic, and tissue-repair signals.
Evidence highlights:
Meta-analysis of MSCs in heart failure: found improved left ventricular ejection fraction (LVEF) and reduced rehospitalization, though no clear reduction in cardiovascular death.
More recent meta-analysis found: “Efficacy of mesenchymal stem cell transplantation on … heart failure caused by ischemic and non-ischemic cardiomyopathy” reported potential for reduced all-cause mortality and improved cardiac function indices.
Here, readers need to understand that meta-analysis is a special kind of studies that combine results of tens of carefully selected studies. Thus, meta-analysis is regarded as highly reliable.
Clinical implication: MSC therapy may serve as a supportive option alongside revascularization, pharmacotherapy, and rehabilitation in heart failure or peripheral ischemia. It may reduce symptoms, improve heart function, and enhance quality of life, though it may or may not reduce heart disease-related mortality.
3. Autoimmune & Inflammatory Conditions
There are more than 100 known autoimmune diseases. At present, there is no cure for these diseases, and most treatments focus on suppressing inflammation. However, these drug therapies have many toxicities, as they suppress immunity and disrupt many metabolic processes. Unlike these drugs (unlike corticosteroids, immunosuppressants, and monoclonal antibodies), stem cell therapy might alter immune responses, changing the course of the disease.
Autoimmune diseases (e.g., multiple sclerosis, systemic lupus erythematosus, Crohn’s disease) involve dysregulated immunity, tissue destruction, and chronic inflammation. MSCs have immunomodulatory and anti-inflammatory effects.
Supporting evidence:
Review: “The role and mechanism of mesenchymal stem cells in type 1 diabetes mellitus” summarizes immunotherapy mechanisms in T1DM but has relevance for autoimmune states.
Review: “Clinical application of mesenchymal stem cells in regenerative medicine” includes immune disease contexts.
Clinical implication: In autoimmune conditions, MSC therapy may be considered as part of a broader immunomodulatory regimen (alongside immunosuppressants, biologics).
4. Multi-Organ & Systemic Metabolic Repair
Beyond targeted organs, MSCs are being trialed in conditions where systemic inflammation and metabolic dysfunction affect multiple organs — e.g., non-alcoholic fatty liver disease, chronic kidney disease, and metabolic syndrome.
Evidence base: There are fewer large trials in these areas; many are preclinical or early-phase. But the underlying mechanisms — MSCs improving mitochondrial function, reducing oxidative stress, and enhancing tissue perfusion — suggest potential.
Clinical implication: For patients with multi‐system involvement of chronic disease (e.g., diabetes with kidney + liver involvement), MSC therapy may offer adjunctive benefit.
Key Themes & Practical Considerations
Safety profile: Across many trials, MSC therapy has been well tolerated with a low incidence of serious adverse events.
Heterogeneity: Major challenges include variations in cell source (bone marrow, adipose, umbilical), dosage, delivery route, patient selection, and outcome measures.
Durability & repeat dosing: Some benefits diminish over time; repeat administrations or combination therapies may be required.
Adjunct nature: Given current evidence, MSCs are best viewed as complementary to standard disease-modifying treatments (medications, surgery, lifestyle) rather than sole therapy.
Regulatory compliance: Many applications are investigational; patients should seek treatment in regulated clinical settings or accredited regenerative medicine centers.
The Bottom Line
For patients living with chronic conditions — whether diabetes, heart disease, arthritis, or autoimmune disorders — MSC therapy presents an exciting frontier. The accumulated evidence suggests that MSCs may reduce inflammation, promote tissue repair, improve function, and support quality of life. Yet, because the field is still maturing, these therapies are not yet substitutes for standard care.
Patients and practitioners interested in MSC therapy should proceed with careful evaluation: verifying cell-product quality, treatment setting credibility, follow-up protocols, and realistic outcome expectations. When used as part of a comprehensive treatment plan, MSCs hold promise to enhance recovery, slow progression, and restore function at the cellular level.
If you want to know how MSC therapy might benefit you, or if it can help manage a chronic health condition you are living with, contact our team or book an appointment.
